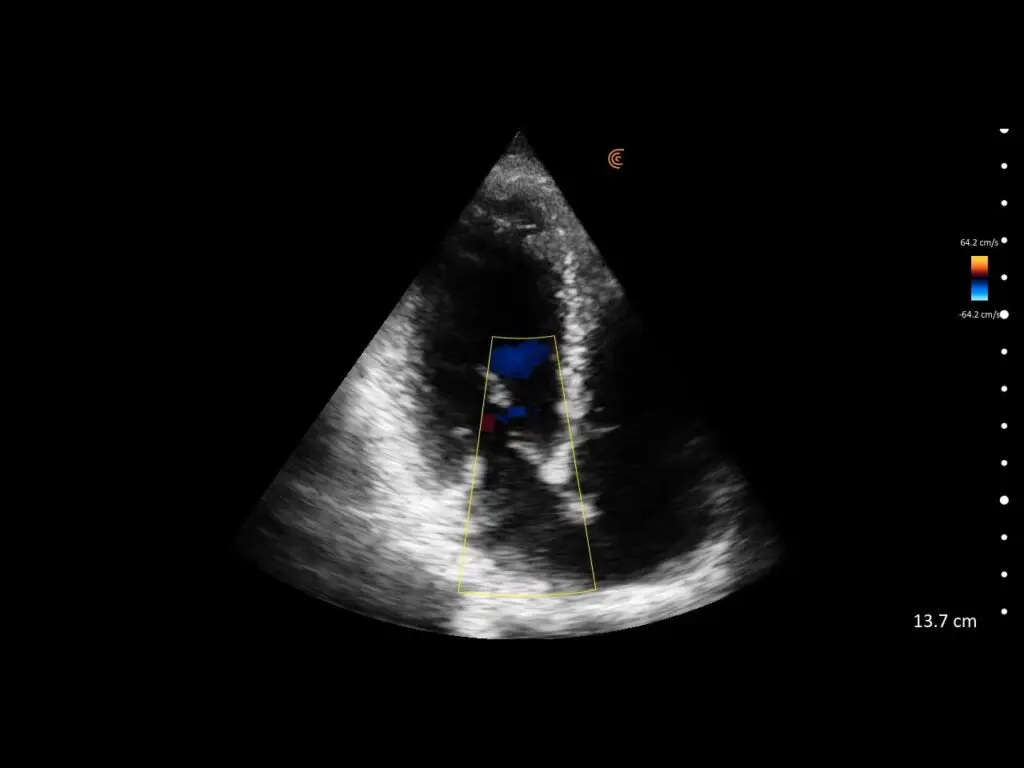
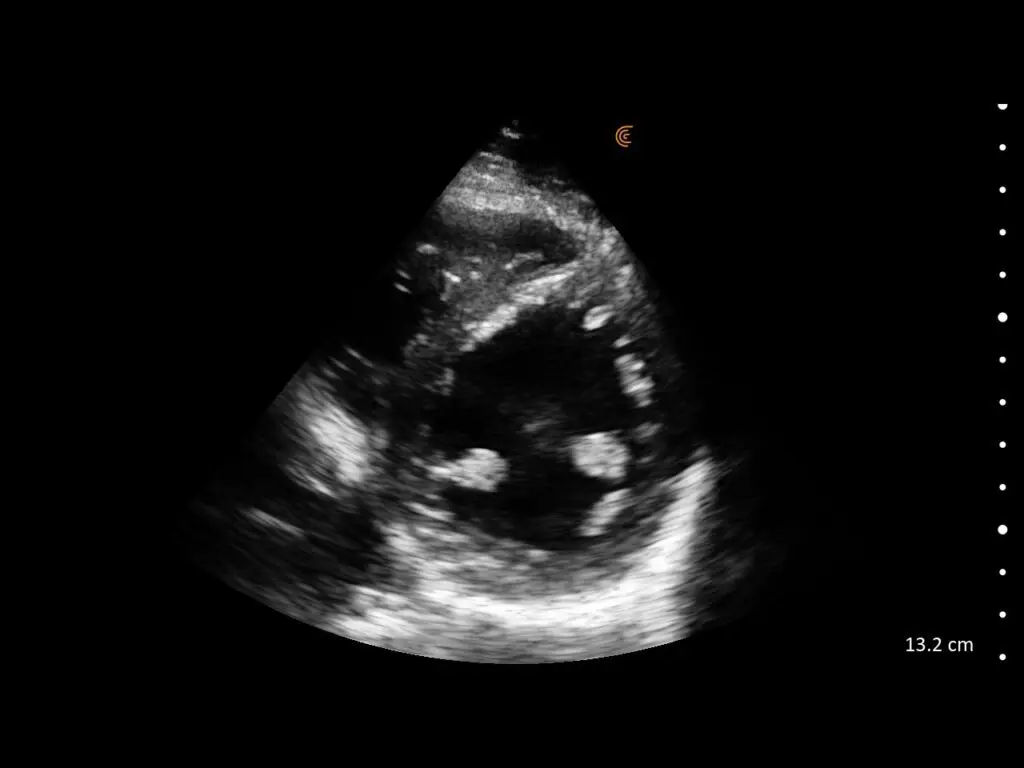
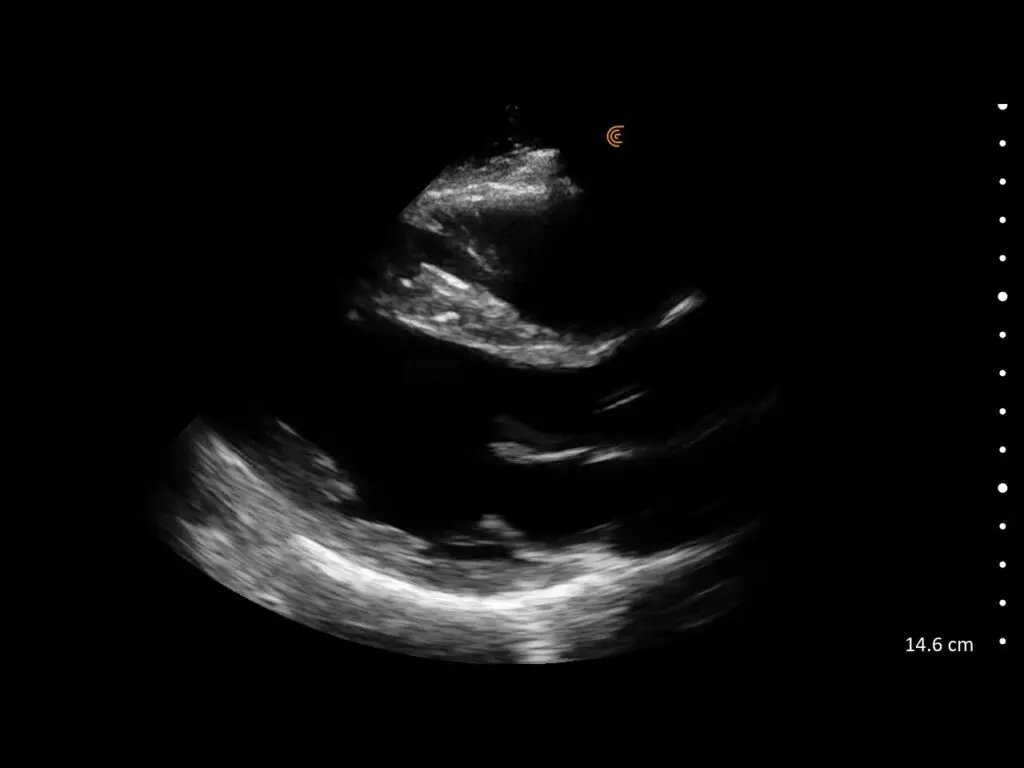
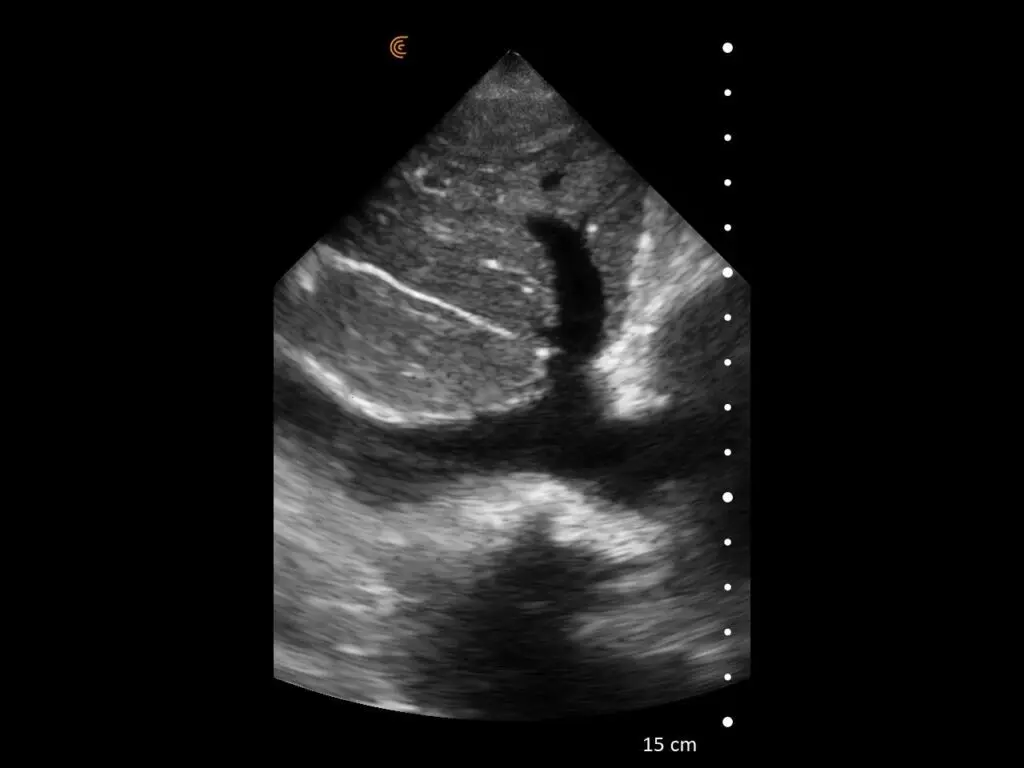
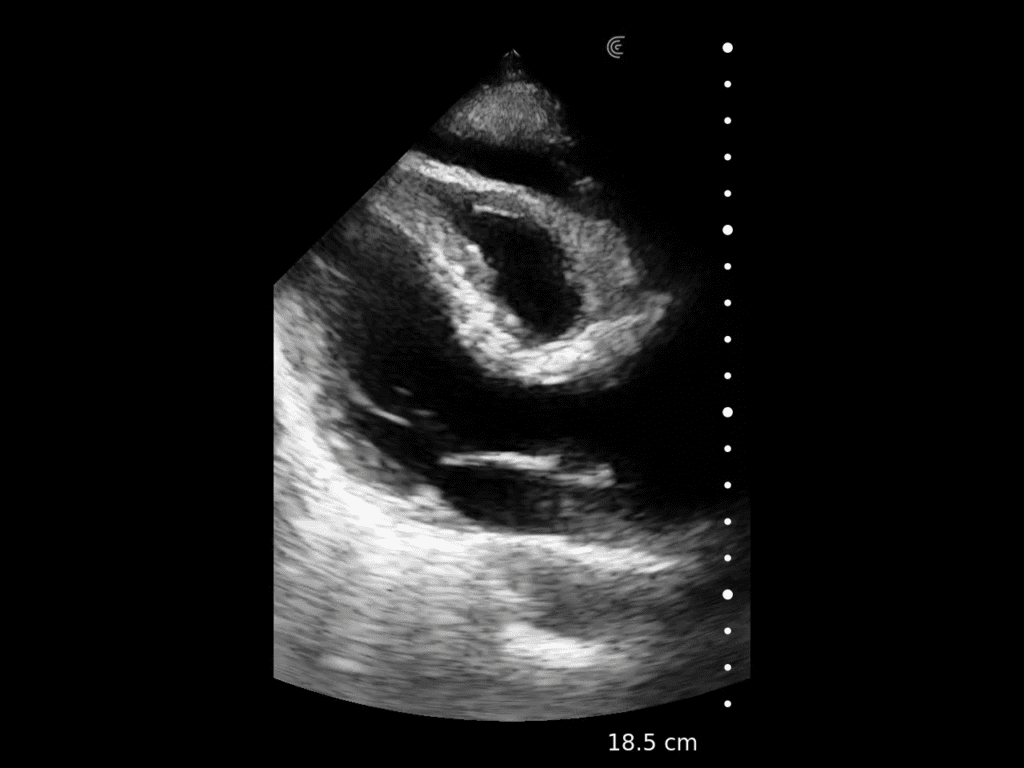
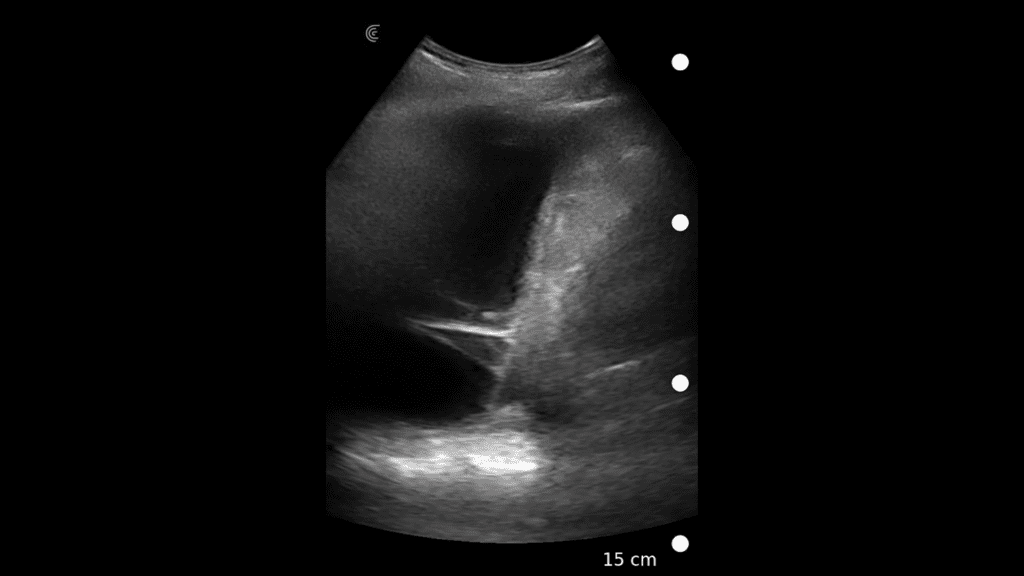
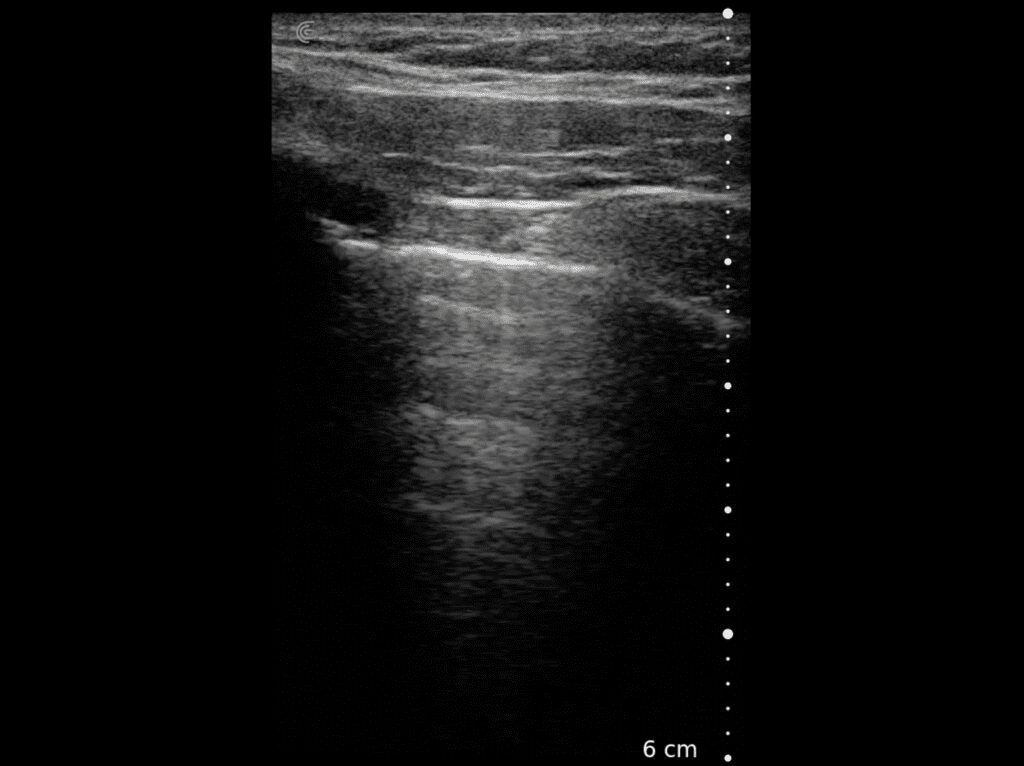
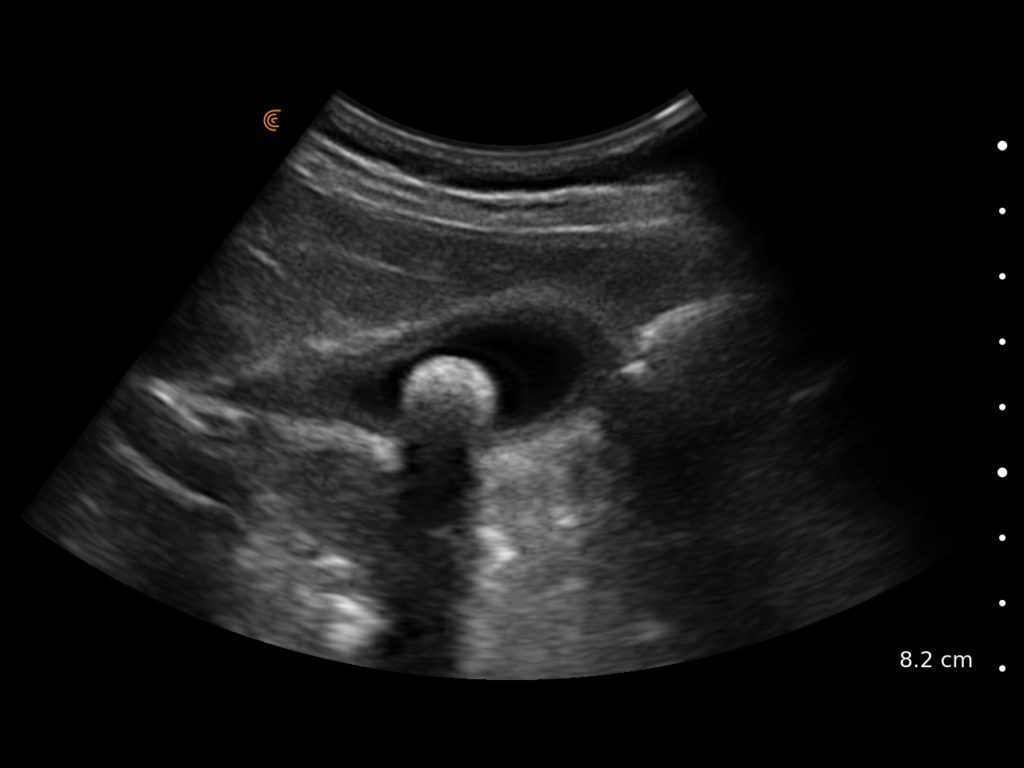
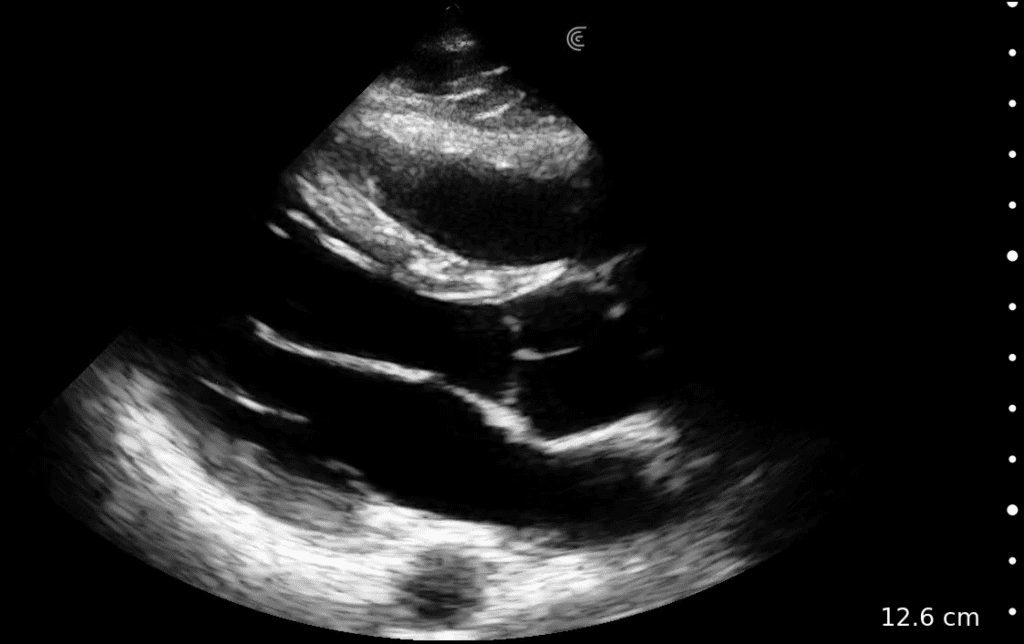
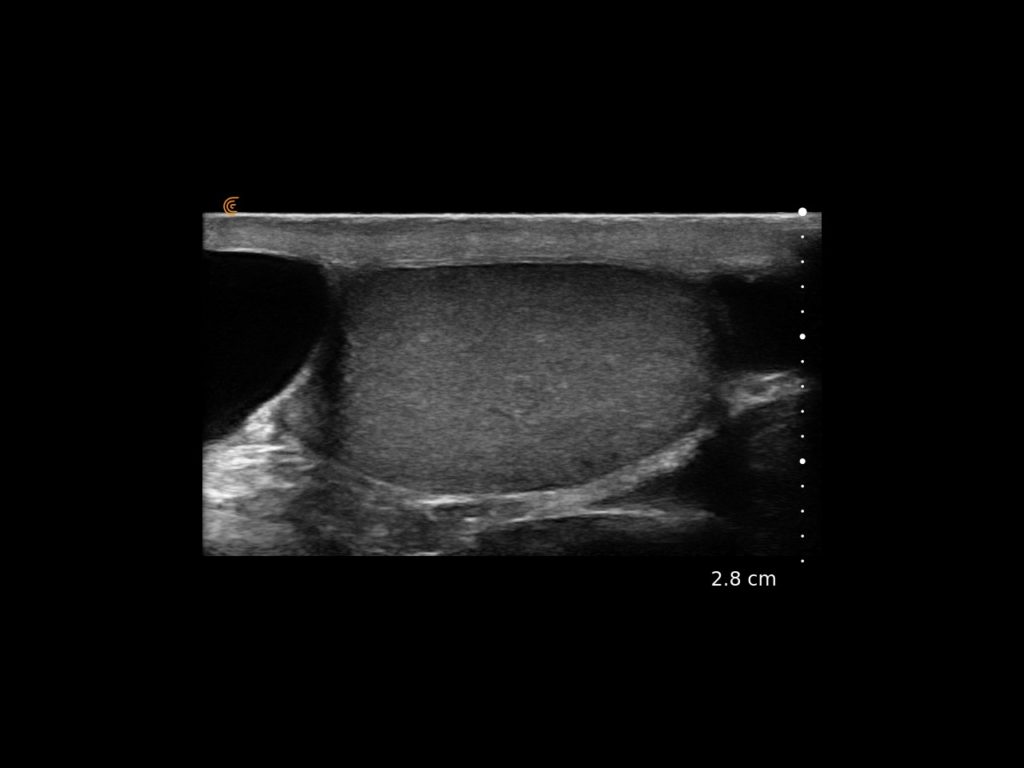
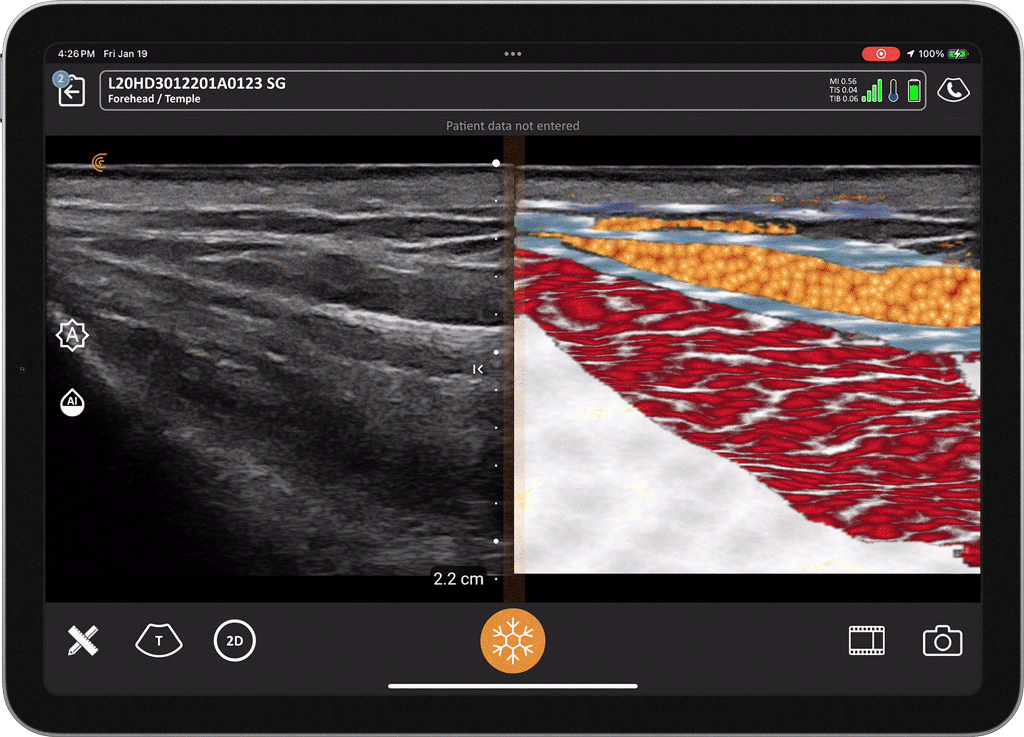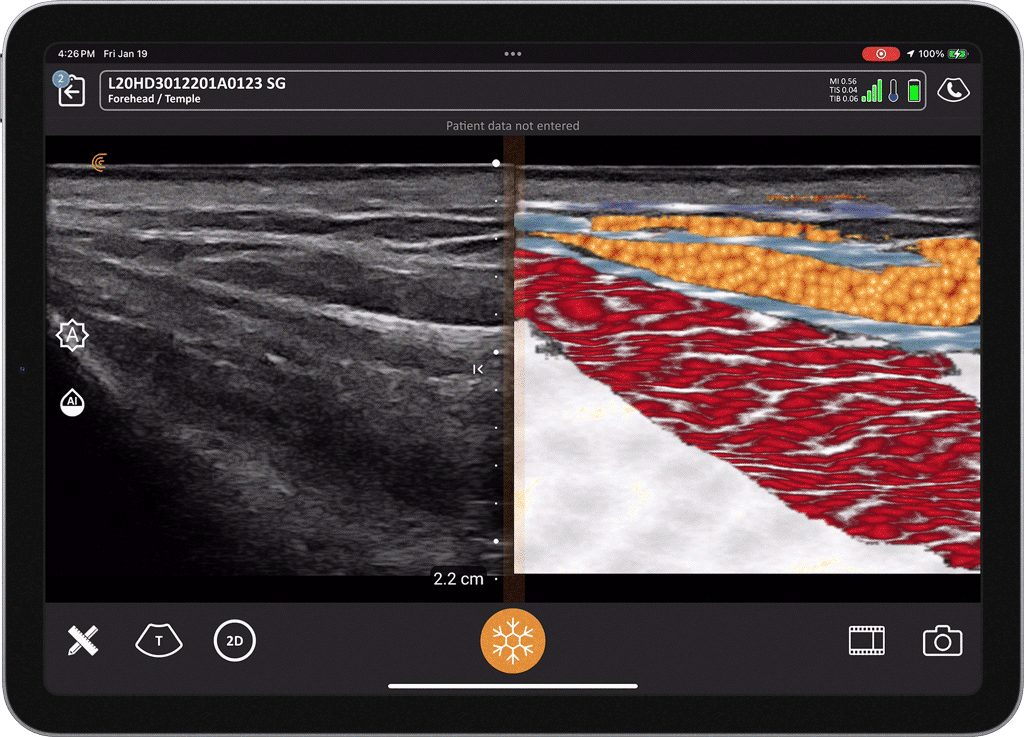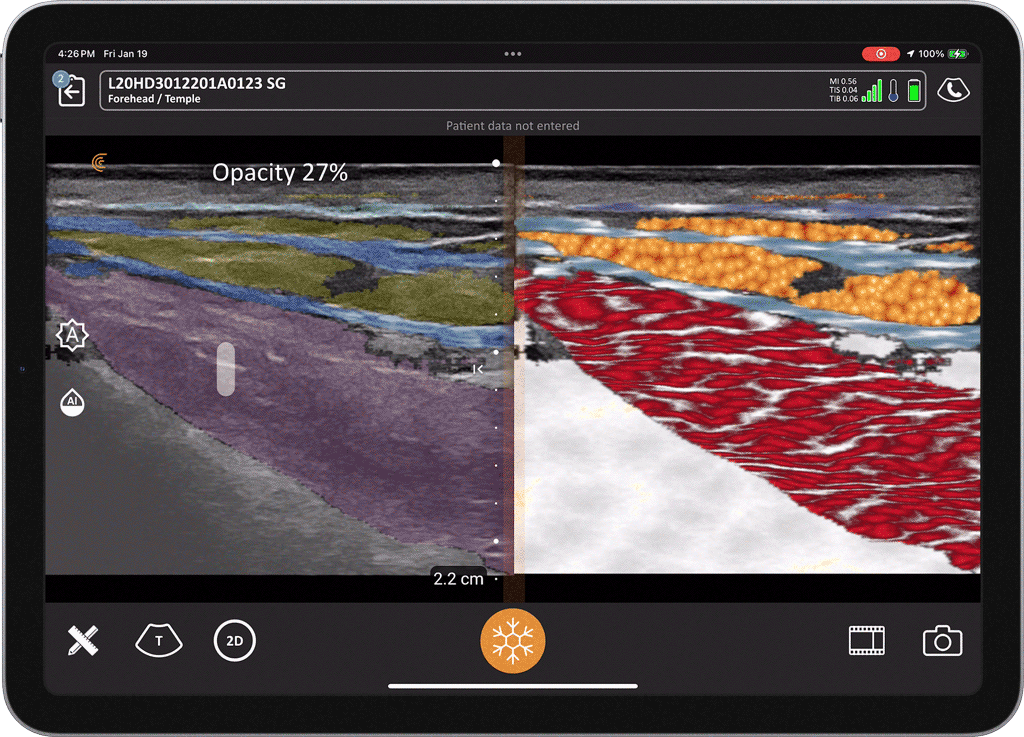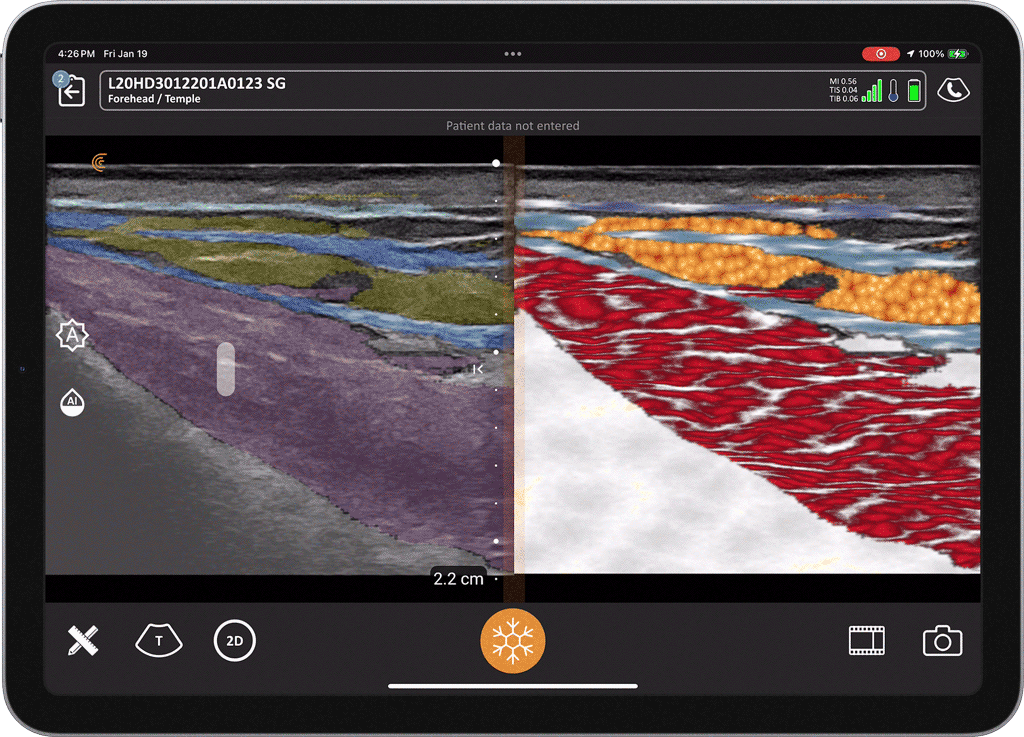
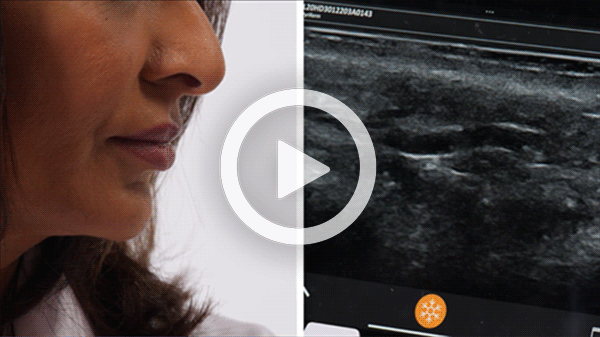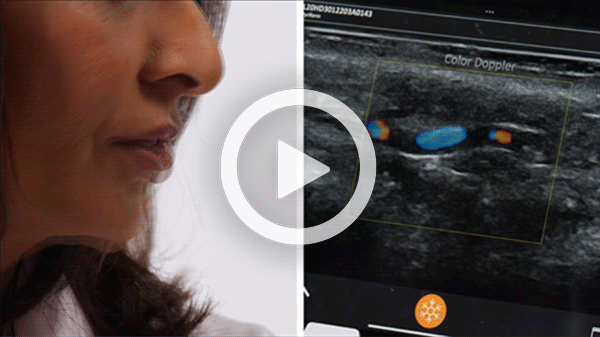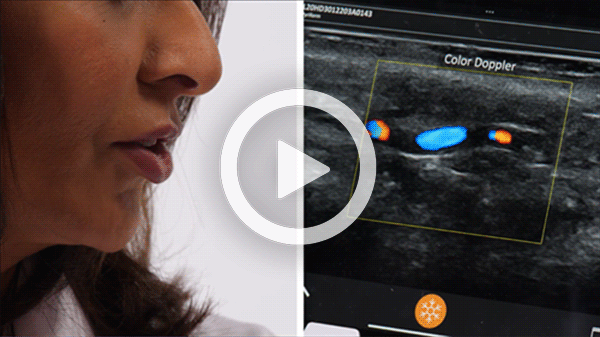
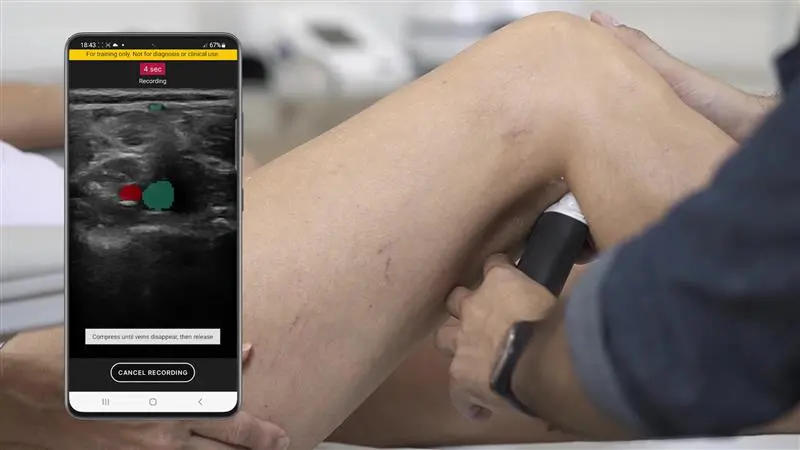
T-Mode is an innovative approach to ultrasound education designed to elevate your scanning skills, using the power of AI. This novel technology overlays distinctive colors, patterns, and labels to instantly identify and differentiate anatomical structures and tissue layers.
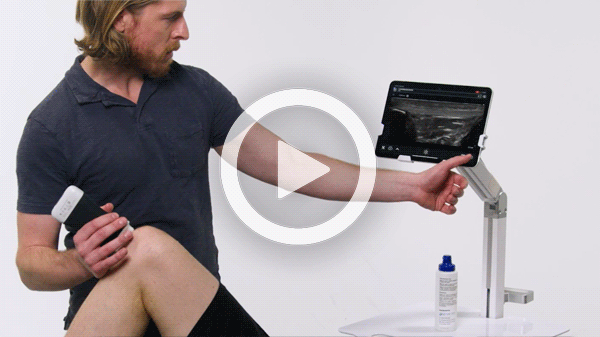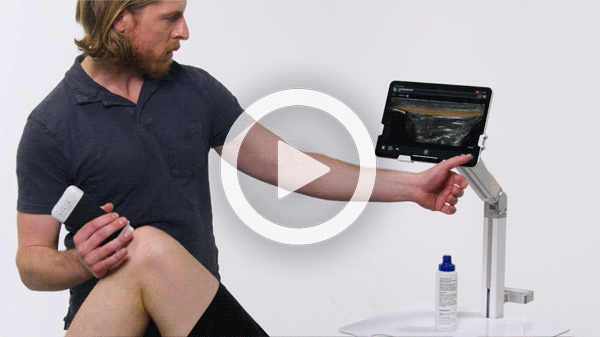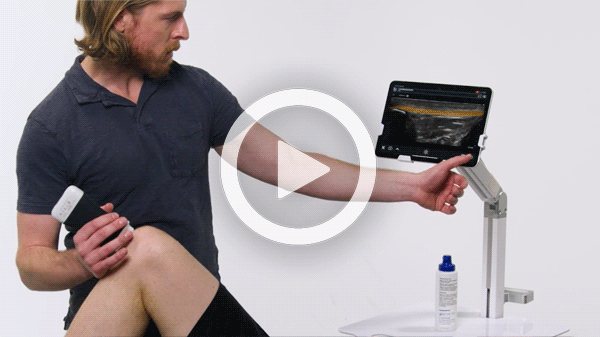
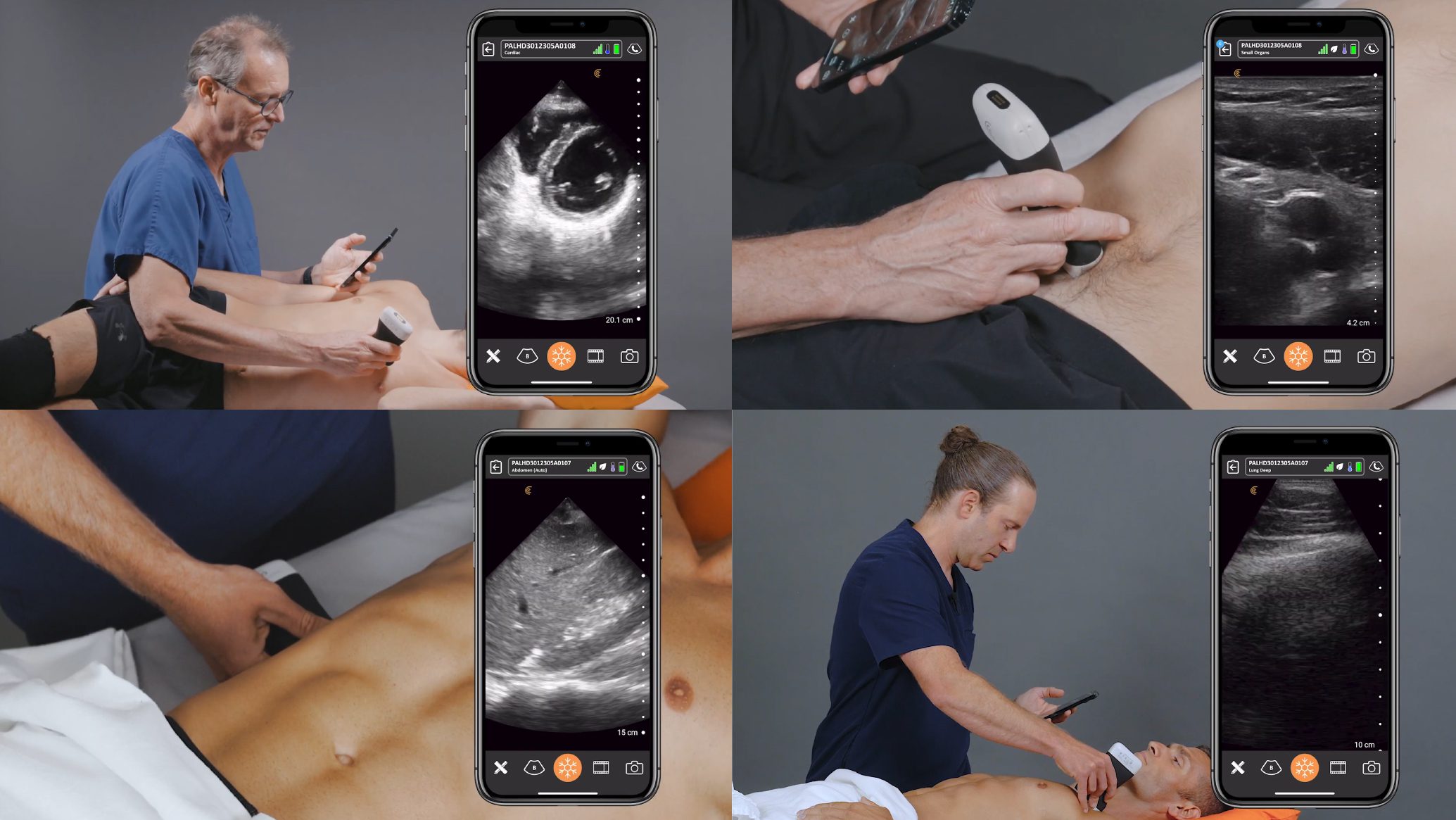
Manage your exams anywhere and improve patient outcomes with clear, real-time imaging that is easy to use, affordable, and ultra-portable.